USP <61> – Microbial Enumeration Test
Get A Testing Quote
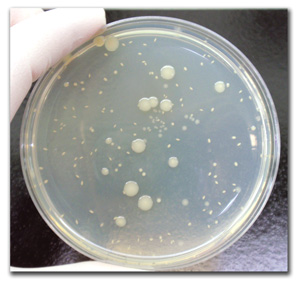
The USP <61> – Microbial Enumeration Test is a product safety test found in Chapter 61 of the United States Pharmacopeia.
It is suggested for use by the FDA for pharmaceuticals as well as cosmetics and personal care products to ensure that a product’s preparation complies with pre-set specifications for microbiological quality.
The USP <61> test is a full quantitative analysis of a product to determine the Total Aerobic Microbial Count (TAMC) and Total Yeast and Mold Count (TYMC) present in the sample. It also includes a neutralization and recovery validation using specific microorganisms. This ensures that the methods used to determine the presence of these microorganisms are appropriate for each individual product.
Similar to an aerobic plate count assay, the USP 61 determines the number of colony-forming units present in a product, however, this method also includes a neutralization and recovery validation using specific microorganisms. While neutralization validation is not required to meet FDA USP <61> criteria, it supports the data and results. The Neutralization/Recoverability Analysis is a control used to show that we can effectively inhibit any antimicrobial agent(s) in the product from killing microorganisms that may have contaminated the product during production. This additional proof showing that the neutralization scheme is effective at inhibiting the active ingredient supports the data collected from the plate counts by showing that the microbial counts reported are an accurate depiction of the microbial content in the sample at the time of testing. We like to say that it provides a “snapshot in time” of the product and proves that these counts are accurate and not inhibited in any way by the recovery scheme.
The pathogens tested in the neutralization validation are opportunistic pathogens whose presence in a cosmetic or personal care product would be a particular concern, especially eye-area cosmetics.
These pathogens include, Staphylococcus aureus, Pseudomonas aeruginosa, and Candida albicans. These three organisms, along with Bacillus subtilis and Aspergillus brasiliensis, are used as indicator organisms for the neutralization and recovery validation portion of the test to show that if these organisms are present in the product at the time of testing, they would be detected during the aerobic plate count portion.
Acceptable Guidelines for Microbial Limits
Cosmetics and personal care products are not expected to be completely free of all microorganisms; however, they must be completely free of high-virulence microbial pathogens, especially in eye-area products, and the total number of microorganisms in the product must be low. Criteria for acceptable counts present in products is based on BAM Chapter 23, a method from the FDA.
For eye-area and baby products, no more than 100 colony forming units (CFU)/ g or ml of product is considered acceptable. For non-eye-area products, no more than 1000 CFU/ g or ml of product is considered acceptable. If the product is determined to have aerobic microbial counts at or below these levels, further testing under USP <62> – Test for Specified Microorganisms – or an equivalent test may be necessary to determine if those microorganisms are high-virulence pathogens.
