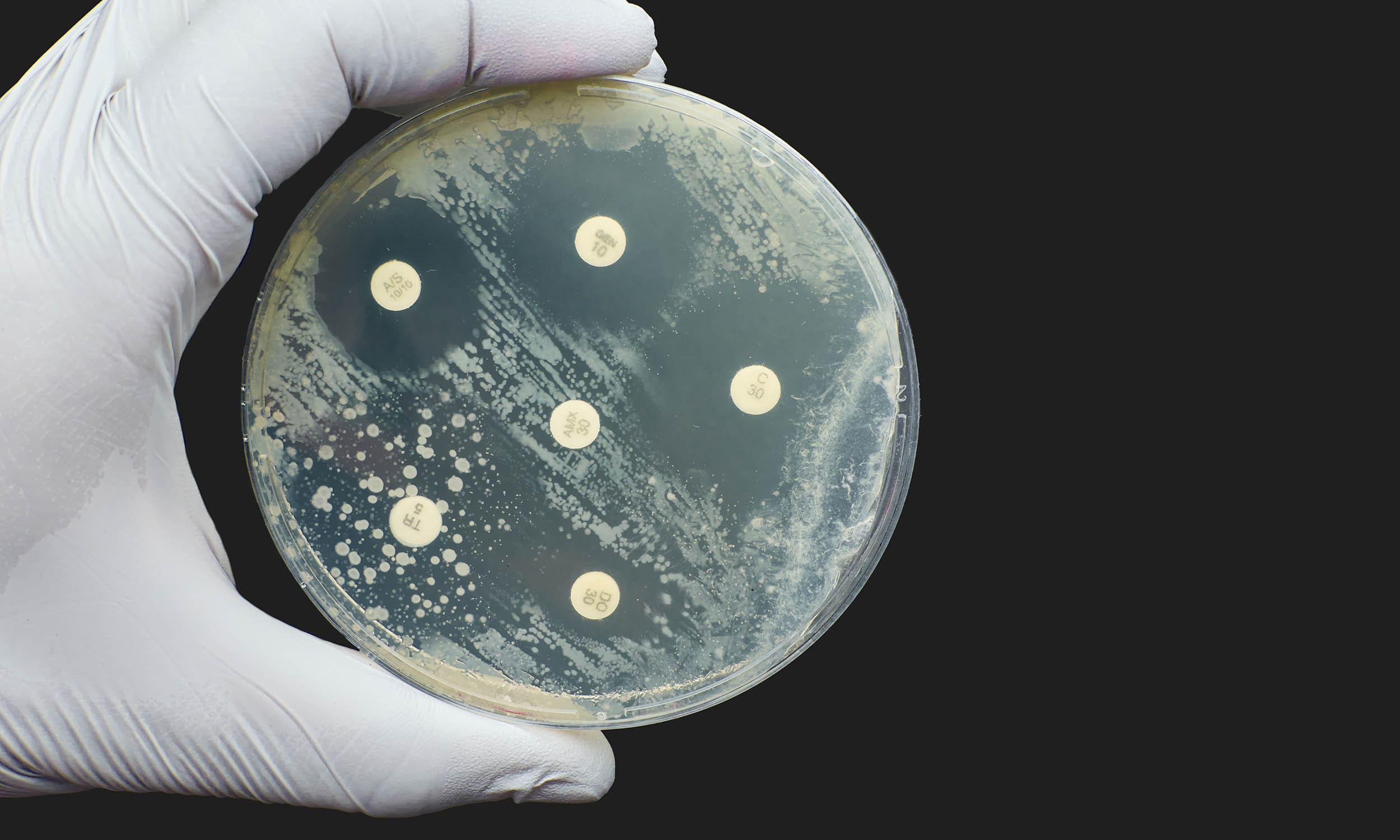
Zone of Inhibition Test, also called the Kirby-Bauer Test
The Zone of Inhibition Test is a qualitative method used clinically to measure antibiotic resistance and industrially to test the ability of solids and textiles to inhibit microbial growth.
This method was originally developed to measure the antibiotic resistance of bacteria or fungi from infected patients. Researchers who develop antimicrobial textiles, surfaces, and liquids now use this test as a quick and easy way to measure antimicrobial activity, particularly the ability of an object or chemical to inhibit microbial growth.
In a zone of inhibition test, approximately one million cells from a single bacterial or fungal strain are spread over an agar plate using a sterile swab then incubated in the presence of the antimicrobial object (such as an oxacillin disk, pictured below). If the bacterial or fungal strain is susceptible to the antimicrobial agent, then a clear zone of resulting from microbial growth inhibition appears on the agar plate. If it is resistant to the antimicrobial agent, then growth is not affected and no clear zone is visible.
Zone of Inhibition Tests are are similar to Minimum Inhibitory Concentration Tests.
Summary of the Zone of Inhibition Test
-
A bacterial or fungal strain of interest is grown in pure culture.
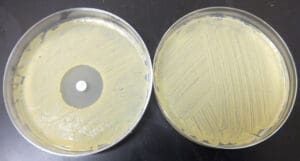
Photograph: Left Plate is of S. aureus with Oxacillin disk, Right Image is lawn growth of S. aureus.
- Using a sterile swab, a suspension of the pure culture is spread evenly over the face of a sterile agar plate.
- The antimicrobial agent is applied to the center of the agar plate (in a fashion such that the antimicrobial doesn’t spread out from the center). A hole can be bored in the center of an agar for a liquid substance.
- The agar plate is incubated for 18-24 hours (or longer if necessary), at a temperature suitable for the test microorganism.
- If antimicrobial agent leaches from the object into the agar and then exerts a growth-inhibiting effect, then a clear zone (the zone of inhibition) appears around the test product.
- The size of the zone of inhibition is usually related to the level of antimicrobial activity present in the sample or product – a larger zone of inhibition usually means that the antimicrobial is more potent.
Strengths of Zone of Inhibition Testing
- Zone of inhibition testing is fast and inexpensive relative to other laboratory tests for antimicrobial activity.
- Zone of inhibition testing is especially well suited for determining (albeit qualitatively) the ability of water-soluble antimicrobials to inhibit the growth of microorganisms.
- A number of samples can be screened for antimicrobial properties quickly using this test method.
- A variety of antimicrobial product types can be tested using this method. Liquids, coated antimicrobial surfaces, and antimicrobial-impregnated solid products can all be tested for their ability to produce a zone of inhibition.
Weaknesses of Zone of Inhibition Testing
- Antimicrobial agents that leach out of the object and into the aqueous agar matrix, such as silver ions, usually show better results than antimicrobials that stay affixed to the object or textile or that are not water-soluble.
- Zone of Inhibition tests do not necessarily indicate that microorganisms have been killed by an antimicrobial product – just that they have been prevented from growing.
- Microbial growth agars themselves may interfere with the function of some antimicrobial agents.
- The method cannot be used to test the activity of antimicrobial agents against viruses, since viruses don’t “grow” on agar plates like bacteria (viruses don’t replicate outside of their host organisms).
- The method has some natural variability, and zones of microbial inhibition do not always have clear or regular boundaries.
- The method is not classically quantitative (though sometimes the diameter of the zones of inhibition are measured and recorded).
Zone of Inhibition Testing is a fast, qualitative means to measure the ability of an antimicrobial agent to inhibit the growth of microorganisms. In the world of antimicrobial substances/surfaces, the degree to which these materials are inhibitory can be of vital importance to the health of the consumer. This test is an outstanding qualitative way for manufacturers of antimicrobial surfaces/substances to be able to compare the inhibition levels of their products.
Contact Microchem for Antimicrobial Testing
Microchem Laboratory provides a full range of antimicrobial tests to support product developers. If your company is interested in screening chemicals, products, or antimicrobial treatments for their ability to inhibit the growth of microorganisms, the zone of inhibition test may be a great place to start. Contact the lab or get a price quote today.