Disinfection Validation Studies

Get A Testing Quote
The EPA requires manufacturers of disinfectants to prove efficacy prior to sale. Nevertheless, the FDA expects critical processes that rely on disinfectants to be validated.
Microchem offers disinfection validation studies for cleanrooms and medical device reprocessing.
Competencies:
- FDA
- Validations
- Study Design
Manufacturers of medical devices and pharmaceuticals that maintain sterile or near-sterile environments, such as cleanrooms, are required by the FDA to validate cleaning and disinfection procedures. In addition, medical device makers are required to validate disinfection procedures for products like blood glucose meters.
There are two main types of tests done for disinfectants in aseptic manufacturing environments:
Medical device manufacturers typically utilize the following:
- Validation of Medical Device Disinfection or Reprocessing.
Current FDA guidance suggests that validation studies should be as realistic as possible. For cleanrooms and aseptic manufacturing environments, studies should include surface types found within the facility as well as recent isolates from facility monitoring programs. For medical devices, disinfection validation studies should look at “worst case” scenarios to prove devices can be reliably disinfected by end-users when instructions are followed.
The FDA requires drug manufacturers to validate disinfection procedures on a facility-by-facility basis, because facilities vary in terms of products used, surface types and predominant microbial contaminants. This page describes the scope and size of a typical disinfection validation study.
Microchem Laboratory has a great deal of experience with regard to design, conduct, and reporting of disinfectant validation studies. The laboratory is FDA GLP-compliant and our customers benefit from our unrivaled focus on disinfectant and sanitizer testing. To start your project with us, contact the lab.
Basic Requirements of a Good Disinfection Validation Study
- The study is designed and run under GLP test conditions.
- A standard test method, such as a modified AOAC Germicidal Spray Test or modified ASTM E 1153 is chosen for use in the study.
- Disinfectants, sterilants, and sporicides used in the facility are tested.
- Surfaces representative of those found in the facility are used in the study.
- A broad range of microorganisms is tested, including “standard” cultures such as S. aureus, E. coli, P. aeruginosa, and A. niger.
- An additional set of microorganisms is tested, representing common environmental isolates. Typically, three isolates are chosen. Common problematic contaminants include Micrococcus species, Bacillus species, and Pseudomonas species.
- Each individual validation test is run in triplicate (three individual replicates).
Table Summarizing a Moderate to Large Disinfection Validation Study
As you may have guessed from reading the “basic requirements” section above, disinfection validation studies can get quite large. The table below summarizes the testing involved in a moderate to large disinfection validation study. Such a study would be appropriate to a large pharmaceutical or medical device manufacturing facility.
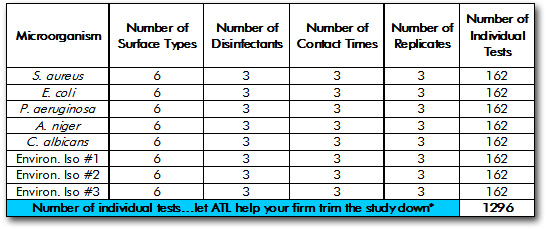
*Microchem Laboratory would hate to see your firm run a study such as the one outlined above in its entirety – it’s just not necessary to do all of those iterations. If your firm needs to contract a disinfection validation study, give us a call or use our contact form to discuss scientifcally defensible strategies to reduce the study size.
Customization of Disinfection Validation Studies
The essence of a disinfection validation study is a reflection in the study design of the sanitation conditions and processes used at a particular facility. Accordingly, Microchem Laboratory works hand in hand with drug makers to ensure that our testing protocols adequately mimic facility disinfection and sanitization. We use a facility’s surface type, disinfectants, and even facility-specific microbial isolates when available.
Share!